-
Phone
+86-0755 2308 4243
-
Address
Room 309, Meihua Building, Taiwan Industrial Park, No.2132 Songbai Road, Bao'an District, Shenzhen, China
-
E-mail
What Is peptide synthesis
Peptide synthesis is an active field in protein and peptide chemistry, which typically involves the sequential addition of amino acids in a defined order to form the peptide chain by chemical methods. The main synthesis methods are liquid-phase and solid-phase synthesis. Compared to LPPS, SPPS has the advantages of being faster, simpler, with fewer side reactions, and higher yields.
Standard SPPS method, including Boc (benzyl )and Fmoc (tBu ) strategies .
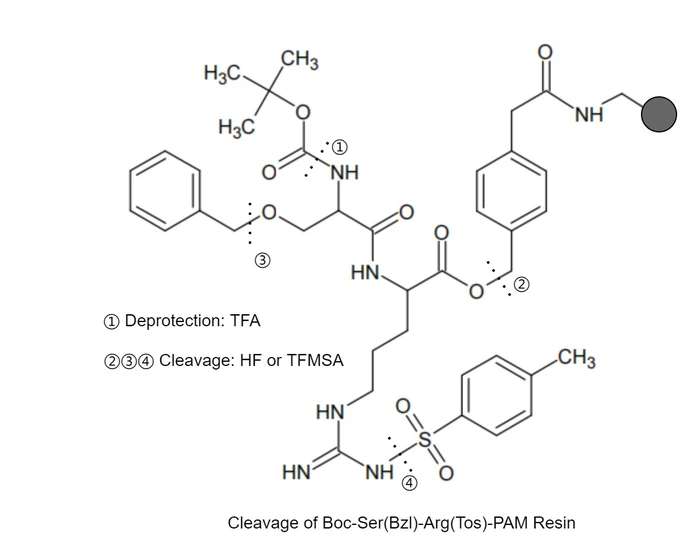
Boc (benzyl ) strategy
In this synthesis approach, Chloromethylated, hydroxymethylated polystyrene and p-methoxybenzyl (PMA) resins are used as solid supports. The α-amino group is protected with t-butoxycarbonyl (Boc), while side chain groups of amino acids are protected with benzyl derivatives. The Boc group is removed using neat TFA or TFA in CH2Cl2, and at the end of the synthesis, side-chain protecting groups and peptide-resin linkages are removed with a strong acid like anhydrous hydrofluoric acid (HF)or TFMSA.
Fmoc (tBu ) strategy
In this synthesis approach, Wang-resin, 2-Chlorotrityl Chloride Resin, Rink amide-AM Resin, Rink amide-MBHA resins are used as solid supports.The α-amino group is protected with 9-fluorenylmethoxycarbonyl (Fmoc), while side chain groups of amino acids are protected with acid-labile protecting groups. The Fmoc group is removed using 20% piperidine in DMF, and at the end of the synthesis, side-chain protecting groups and peptide-resin linkages are removed with 1%-95% TFA. Fmoc SPPS is currently the preferred method for peptide synthesis.

Basic Principles of Fmoc Solid-phase Peptide Synthesis
Choose Resin
The most widely applied resins in Fmoc SPPS including 4-Alkoxybenzyl alcohol (Wang) resin, 2-Chlorotrityl chloride resin, Rink Amide Resin, Rink Amide-MBHA resins. Amide resins are used for synthesis peptides with requried C terminal amidation. Wang resin and 2-Cl Trt resin are commonly used for synthesis peptide with requried C terminal free COOH. But when the C-terminus of the peptide has Pro and Gly as the first amino acid, Wang resin should not be used due to the risk of DKP (diketopiperazine) formation, 2-Chlorotrityl Chloride Resin is recommended for use.
 |
 |
Attachment of protected amino acids to Resins
The first Fmoc amino acid is attached to an insoluble support resin via an acid labile linker.
Deprotection of the α-amino protecting group
The temporary Fmoc protecting group at the N-terminus of the peptidyl-resin is removed by treating it with a solution of 20% piperidine in DMF. This reaction usually finishes within 10 to 20 minutes.
Washing steps
After each chemical reaction, washing steps are performed to remove excess reagents, by-products, and unreacted molecules from the resin. This helps to maintain the purity of the growing peptide chain.
Activation and coupling of the protected amino acid
The protected amino acid bound to the resin is activated to enhance its reactivity for coupling with the next amino acid in the sequence. Common activating agents include HOAt, HOBt, PyBOP, PyAOP, HBTU and HATU . Coupling involves forming a peptide bond between the activated amino acid and the amino group of the growing peptide chain. A Kaiser test is typically used to monitor the completeness of coupling reactions.
Washing steps
Similar to earlier washing steps, this ensures the removal of excess reagents and by-products after the coupling reaction.
Cleavage from resin and deprotection of side chain protecting groups
Once the desired peptide sequence is synthesized on the resin, it is cleaved from the solid support using a cleavage cocktail such as trifluoroacetic acid (TFA) containing scavengers like thioanisole and water. Simultaneously, side chain protecting groups on amino acid residues are removed, revealing the fully deprotected peptide ready for purification and analysis.
