As a seasoned supplier of peptide Active Pharmaceutical Ingredients (APIs), I've witnessed firsthand the pivotal role that high - quality peptide APIs play in the pharmaceutical industry. Peptide APIs are the building blocks of many innovative drugs, and their quality can significantly impact the safety, efficacy, and overall success of the final pharmaceutical products. In this blog, I'll delve into the various factors that affect the quality of peptide APIs.
Raw Material Quality
The quality of raw materials is the cornerstone of high - quality peptide APIs. Amino acids, the basic units of peptides, must meet strict purity and quality standards. Impurities in amino acids can lead to the formation of unwanted by - products during peptide synthesis. For example, if an amino acid contains trace amounts of isomers or contaminants, these can be incorporated into the growing peptide chain, resulting in a heterogeneous product mixture.
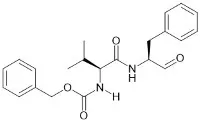
Moreover, the source of raw materials also matters. We source our amino acids from reliable suppliers who adhere to Good Manufacturing Practices (GMP). This ensures that the amino acids are produced under controlled conditions, with proper documentation of their origin, production process, and quality control measures. For instance, when synthesizing a peptide API that requires a specific protected amino acid like Fmoc - L - Lys[C20 - OtBu - γ - Glu(OtBu) - AEEA - AEEA] - OH CAS 2915356 - 76 - 0, we carefully select suppliers who can guarantee its high purity and stability.
Synthesis Process
The synthesis process of peptide APIs is a complex and multi - step procedure. There are two main methods for peptide synthesis: solid - phase peptide synthesis (SPPS) and solution - phase peptide synthesis. Each method has its own advantages and challenges, and the choice of method can affect the quality of the final product.
SPPS is widely used in the industry due to its simplicity, efficiency, and ease of automation. However, it requires careful control of reaction conditions, such as the coupling time, temperature, and the amount of reagents. Inadequate coupling can lead to incomplete peptide chain elongation, resulting in truncated peptides. On the other hand, over - reaction can cause side reactions, such as racemization or the formation of peptide aggregates.
In solution - phase peptide synthesis, the reaction occurs in a homogeneous solution. This method allows for better control of reaction kinetics and can be used for the synthesis of large peptides. However, it often involves multiple purification steps, which can increase the risk of product loss and contamination.
We have developed a set of optimized synthesis protocols for different peptide APIs. For example, when synthesizing Semaglutide CAS 910463 - 68 - 2, a popular GLP - 1 receptor agonist, we carefully adjust the reaction parameters based on the specific characteristics of the peptide sequence to ensure high - yield and high - quality synthesis.
Purification Techniques
Purification is a crucial step in ensuring the quality of peptide APIs. Crude peptides synthesized in the laboratory usually contain various impurities, such as truncated peptides, by - products, and residual reagents. These impurities need to be removed to obtain a pure and homogeneous peptide API.
Common purification techniques include high - performance liquid chromatography (HPLC), ion - exchange chromatography, and size - exclusion chromatography. HPLC is the most widely used method due to its high resolution and selectivity. It can separate peptides based on their hydrophobicity, charge, or size. However, the choice of stationary phase, mobile phase, and elution conditions in HPLC can significantly affect the purification efficiency.
We use state - of - the - art purification equipment and advanced purification strategies. For example, we may use a combination of different chromatography methods to achieve a high - purity peptide API. When purifying Boc - His(Boc) - Aib - OH CAS 1169630 - 98 - 1, we first use ion - exchange chromatography to remove charged impurities and then use HPLC for final purification to obtain a product with a purity of over 95%.
![Fmoc-L-Lys[C20-OtBu-γ-Glu(OtBu)-AEEA-AEEA]-OH CAS 2915356-76-0](/uploads/42783/fmoc-l-lys-c20-otbu-glu-otbu-aeea-aeea-ohf83ab.png)

Quality Control and Testing
Quality control and testing are essential throughout the production process of peptide APIs. From raw material inspection to the final product release, a series of tests are carried out to ensure that the peptide APIs meet the required quality standards.
Physical and chemical properties such as molecular weight, purity, and amino acid composition are routinely tested. Mass spectrometry is used to determine the molecular weight of the peptide, while HPLC is used to measure its purity. Amino acid analysis can confirm the correct amino acid composition of the peptide.
In addition, biological activity tests are also necessary for peptide APIs used in therapeutic applications. These tests can evaluate the ability of the peptide to interact with its target molecule and exert the desired biological effect. For example, for a peptide API that acts as a receptor agonist, in vitro receptor binding assays and cell - based functional assays can be used to assess its biological activity.
We have a dedicated quality control laboratory equipped with advanced analytical instruments. Our quality control team follows strict standard operating procedures (SOPs) to ensure the accuracy and reliability of the test results.
Storage and Handling
Proper storage and handling are also important factors affecting the quality of peptide APIs. Peptides are sensitive to environmental factors such as temperature, humidity, and light. Improper storage can lead to peptide degradation, aggregation, or loss of biological activity.
Peptide APIs should be stored at low temperatures, usually at - 20°C or below. They should also be protected from moisture and light. When handling peptide APIs, clean and dry equipment should be used to avoid contamination.
We provide detailed storage and handling instructions to our customers. We also ensure that our products are properly packaged and shipped under controlled conditions to maintain their quality during transportation.
Regulatory Compliance
Regulatory compliance is a key factor in the production of peptide APIs. Pharmaceutical products, including those containing peptide APIs, are subject to strict regulations in different countries and regions. Compliance with regulations such as GMP, Good Laboratory Practices (GLP), and Good Clinical Practices (GCP) is essential to ensure the safety and efficacy of peptide APIs.
We are committed to meeting all relevant regulatory requirements. Our manufacturing facilities are regularly inspected and certified to ensure that they comply with international standards. This not only guarantees the quality of our peptide APIs but also gives our customers confidence in our products.
In conclusion, the quality of peptide APIs is affected by multiple factors, including raw material quality, synthesis process, purification techniques, quality control and testing, storage and handling, and regulatory compliance. As a peptide API supplier, we are dedicated to controlling these factors to provide high - quality peptide APIs to our customers.
If you are interested in purchasing high - quality peptide APIs, please feel free to contact us for more information and to discuss your specific requirements. We look forward to establishing a long - term and mutually beneficial partnership with you.
References
- Alberts, B., et al. Molecular Biology of the Cell. Garland Science, 2014.
- Gross, E., and Meienhofer, J. The Peptides: Analysis, Synthesis, Biology. Academic Press, 1979 - 1987.
- Chan, W. C., and White, P. D. Fmoc Solid - Phase Peptide Synthesis: A Practical Approach. Oxford University Press, 2000.