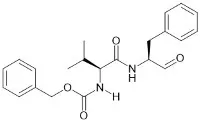
Antibody-drug conjugates (ADCs) have emerged as a promising class of targeted cancer therapeutics, combining the specificity of monoclonal antibodies with the potent cytotoxicity of small molecule drugs. Peptide linkers play a crucial role in ADCs, influencing their pharmacokinetics, stability, and efficacy. As a leading supplier of peptide linkers for ADCs, we understand the significance of these components and their impact on the overall performance of ADCs. In this blog, we will explore how peptide linkers influence the pharmacokinetics of ADCs and discuss the implications for drug development.
Pharmacokinetics of ADCs
Pharmacokinetics (PK) refers to the study of how a drug is absorbed, distributed, metabolized, and excreted by the body. In the context of ADCs, PK is particularly important as it determines the drug's exposure at the target site and its overall safety profile. The PK of ADCs is influenced by several factors, including the antibody, the linker, and the payload.
The antibody component of an ADC provides specificity for the target antigen, allowing the conjugate to selectively bind to cancer cells. The linker connects the antibody to the payload, and its properties can significantly affect the PK of the ADC. The payload is the cytotoxic drug that is released upon internalization of the ADC by the target cells.
Influence of Peptide Linkers on ADC Pharmacokinetics
1. Stability and Circulation Time
One of the key roles of peptide linkers is to maintain the stability of the ADC in the bloodstream. A stable linker ensures that the payload remains attached to the antibody until it reaches the target cells, preventing premature release of the cytotoxic drug and reducing off-target toxicity. Peptide linkers can be designed to be stable under physiological conditions but cleavable in the acidic environment of the endosomes or lysosomes after internalization by the target cells.
For example, our Acetylene - linker - Val - Cit - PABC - MMAE is a cleavable peptide linker that is stable in circulation but can be efficiently cleaved by lysosomal enzymes. This stability allows the ADC to circulate in the bloodstream for an extended period, increasing the chances of reaching the target cells and delivering the payload.
The length and composition of the peptide linker can also affect the circulation time of the ADC. Longer linkers may increase the flexibility of the conjugate, potentially improving its pharmacokinetic properties. However, excessively long linkers may also increase the immunogenicity of the ADC or lead to reduced stability.
2. Tissue Distribution
Peptide linkers can influence the tissue distribution of ADCs by affecting the size, charge, and hydrophobicity of the conjugate. These properties can determine how the ADC interacts with different tissues and cells in the body.
A linker with appropriate hydrophilicity can enhance the solubility of the ADC, preventing aggregation and improving its distribution in the bloodstream. On the other hand, a hydrophobic linker may increase the affinity of the ADC for cell membranes, potentially leading to increased uptake by non - target tissues.
Our Alkyne - Val - Cit - PAB - OH linker is designed to optimize the tissue distribution of ADCs. The alkyne group allows for efficient conjugation to the antibody, while the Val - Cit - PAB moiety provides cleavability and appropriate physicochemical properties for targeted delivery.
3. Metabolism and Clearance
The metabolism and clearance of ADCs are also influenced by the peptide linker. Cleavable linkers can be designed to be degraded by specific enzymes in the body, leading to the release of the payload and subsequent clearance of the antibody and linker fragments.
Non - cleavable linkers, on the other hand, may require the entire ADC to be internalized and degraded by the target cells. The rate of metabolism and clearance can affect the exposure of the payload at the target site and the overall safety of the ADC.
Our Boc - Val - Cit - PAB - OH linker is a precursor for the synthesis of cleavable peptide linkers. The Boc group can be removed during the conjugation process, and the Val - Cit - PAB moiety can be cleaved by lysosomal enzymes, facilitating the release of the payload and the clearance of the ADC components.
Implications for ADC Drug Development
Understanding the influence of peptide linkers on the pharmacokinetics of ADCs is crucial for the development of effective and safe cancer therapeutics. By carefully designing the peptide linker, researchers can optimize the PK properties of the ADC, improving its efficacy and reducing its toxicity.
In drug development, pre - clinical studies are often conducted to evaluate the PK of ADCs with different peptide linkers. These studies can provide valuable information about the circulation time, tissue distribution, metabolism, and clearance of the conjugates. Based on the results of these studies, the most promising peptide linkers can be selected for further development.
In addition, the choice of peptide linker can also impact the manufacturing process of ADCs. Linkers that are easy to synthesize and conjugate to the antibody can simplify the production process and reduce costs.
Conclusion
Peptide linkers play a vital role in influencing the pharmacokinetics of ADCs. Their properties, such as stability, length, composition, and cleavability, can significantly affect the circulation time, tissue distribution, metabolism, and clearance of the conjugates. As a supplier of peptide linkers for ADCs, we are committed to providing high - quality linkers that can help researchers optimize the PK properties of their ADCs.
If you are interested in learning more about our peptide linkers for ADCs or are looking to discuss potential procurement opportunities, we encourage you to reach out to us. Our team of experts is available to provide detailed information and support to help you in your drug development projects.
References
- Ducry, L., & Stump, B. (2010). Antibody-drug conjugates: linking cytotoxic payloads to monoclonal antibodies. Bioconjugate Chemistry, 21(1), 5 - 13.
- Carter, P. J., & Senter, P. D. (2008). Antibody-drug conjugates for cancer therapy. Cancer Journal, 14(3), 154 - 169.
- Lyon, R. P., & Clynes, R. (2012). Antibody-drug conjugates: targeted delivery of cytotoxic agents. Annual Review of Pharmacology and Toxicology, 52, 171 - 197.